JEE Advanced | 2015Work, Power and EnergyHard
Question
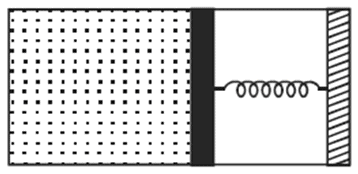
An ideal monoatomic gas is confined in a horizontal cylinder by a spring loaded piston (as shown
in the figure). Initially the gas is at temperature T1, pressure P1 and volume V1 and the spring is in
its relaxed state. The gas is then heated very slowly to temperature T2, pressure P2 and volume V2.
During this process the piston moves out by a distance x. Ignoring the friction between the piston
and the cylinder, the correct statement(s) is(are) :-
in the figure). Initially the gas is at temperature T1, pressure P1 and volume V1 and the spring is in
its relaxed state. The gas is then heated very slowly to temperature T2, pressure P2 and volume V2.
During this process the piston moves out by a distance x. Ignoring the friction between the piston
and the cylinder, the correct statement(s) is(are) :-

Options
A.If V2 = 2V1 and T2 = 3T1, then the energy stored in the spring is 1/4 P1V1
B.If V2 = 2V1 and T2 = 3T1, then the change in internal energy is 3 P1V1
C.If V2 = 3V1 and T2 = 4T1, then the work done by the gas is 7/3 P1V1
D.If V2 = 3V1 and T2 = 4T1, then the heat supplied to the gas is 17/6 P1V1
Solution
if V2 = 2V1 & T2 = 3T1
⇒ P2 =
ᐃU = f/2 (nRᐃT) =
=
Create a free account to view solution
View Solution FreeMore Work, Power and Energy Questions
The upper half of an inclined plane with inclination φ is perfectly smooth while the lower half is rough. A body st...In the arrangement of 3 bulbs of (50W, 100V) each as shown in the figure total power consumption will be :-...A ball ′A′ moving with a speed of 90 ms−1 collides directly with another identical ball ′B′...A Carnot engine, having an efficiency of as heat engine, is used as a refrigerator. If the work done on the system is 10...A 2 Kg mass lying on a table is displaced in the horizontal direction through 50 cm. The work done by the normal reactio...