Redox and Equivalent ConceptHard
Question
At 298 K, the standard reduction potentials are 1.51 V for MnO4- |Mn2+, 1.36 V for Cl2|Cl- , 1.07 V for Br2|Br, and 0.54 V for I2|I-. At pH = 3, permanganate is expected to oxidize :(RT/F = 0.059 V)
Options
A.Cl-, Br- and I-
B.Br- and I-
C.Cl- and Br-
D.I- Only
Solution
MnO + 8H+ + 5e- → Mn2+ + 4H2O
E = 1.51 -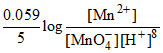
Taking Mn2+ and MnO4- in standard state i.e. 1 M,
E = 1.51 -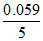 × 8 log
× 8 log 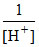
= 1.51 -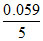 × 8 × 3 = 1.2268 V
× 8 × 3 = 1.2268 V
Hence at this pH, MnO4- will oxidise only Br- and I- as SRP of Cl2/Cl- is 1.36 V which is greater than that for MnO4- /Mn2+.
E = 1.51 -
Taking Mn2+ and MnO4- in standard state i.e. 1 M,
E = 1.51 -
= 1.51 -
Hence at this pH, MnO4- will oxidise only Br- and I- as SRP of Cl2/Cl- is 1.36 V which is greater than that for MnO4- /Mn2+.
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
The volume of 0.10 M-AgNO3 should be added to 10.0 ml of 0.09 M-K2CrO4 to precipitate all the chromate as Ag2CrO4 is...In the following unbalanced redox reaction, Cu3P + Cr2O72− → Cu2+ + H3PO4 + Cr3+, the equivalent weight of H3PO4 is...The number of moles of Cr2O72− needed to oxidize 0.136 equivalent of N2H5+ through the reactionN2H5+ + Cr2O72− → N2 + Cr...Which of the following oxide can not act as reducing agent ?...Preparation of lyphobic sols by chemical method involves...