Aromatic HydrocarbonsHard
Question
A solution at 20oC is composed of 1.5 mol of benzene and 3.5 mol of toluene. If the vapour pressure of pure benzene and pure toluene at this temperature are 74.7 torr and 22.3 torr, respectively, then the total vapour pressure of the solution and the benzene mole fraction in equilibrium with it will be, respectively :
Options
A.35.0 torr and 0.480
B.30.5 torr and 0.389
C.38.0 torr and 0.589
D.35.8 torr and 0.280
Solution
xBenzene = 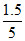 = 0.3, xtoluene = 0.7
= 0.3, xtoluene = 0.7
⇒ PT = 74.7 × 0.3 + 0.7 × 22.3
= 22.41 + 15.61
= 38.02 torr
and ybenzene =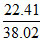 = 0.589
= 0.589
⇒ PT = 74.7 × 0.3 + 0.7 × 22.3
= 22.41 + 15.61
= 38.02 torr
and ybenzene =
Create a free account to view solution
View Solution FreeMore Aromatic Hydrocarbons Questions
Electrophilic attack of NO2⊗ at meta position is observed in :...Anhydrous AlCl3 is used in the Friedel − Craft′s reaction because it is -...Benzene reacts with sulphuric acid to form benzenesulphonic acid, only when the sulphuric acid is-...Friedel−Crafts′ reaction does not occur in case of -...Which product is/are obtained in following reaction....