Chemical BondingHard
Question
Arrange the following amines in the order of increasing basicity :
Options
A.CH3 NH2 < 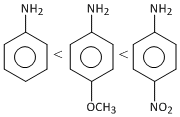

B.  < CH3 NH2
< CH3 NH2
 < CH3 NH2
< CH3 NH2C. 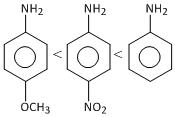 < CH3 NH2
< CH3 NH2
 < CH3 NH2
< CH3 NH2D. 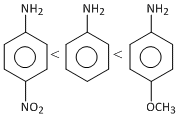 < CH3 NH2
< CH3 NH2
 < CH3 NH2
< CH3 NH2Solution
CH3-NH2 pKa = 10.64
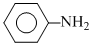 pKa = 4.62
pKa = 4.62
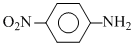 pKa = 0.98
pKa = 0.98
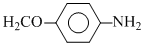 pKa = 5.29
pKa = 5.29
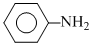 pKa = 4.62
pKa = 4.62 pKa = 0.98
pKa = 0.98 pKa = 5.29
pKa = 5.29Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Find a set of species which consists of only paramagnetic species...Which of the following solid has maximum melting point :-...Which contains both polar and mom-polar bonds ?...Which of the following is/are disproportionation reaction ?...The geometry of CIO3- according to active to valence shell electron pair repulsion (VSEPR)theory will be...