Alkyl Halides and Aryl HalidesHard
Question
For the first order reaction
2N2O5 (g) → 4NO2(g) + O2(g)
2N2O5 (g) → 4NO2(g) + O2(g)
Options
A.the concentration of the reactant decreases exponentially with time
B.the half-life of the reaction decreases with increasing temperature
C.the half-life of the reaction depends on the initial concentration of the reactant
D.the reaction proceeds to 99.6% completion in eight half-life duration
Solution
For first order reaction
[A] = [A]0e-kt
Hence concentration of [NO0] decreases exponentially.Also, t1/2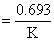 . Which is independent of concentration and t1/2 decreases with the increase of temperature.
. Which is independent of concentration and t1/2 decreases with the increase of temperature.
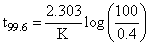
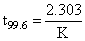 log (2.4) = 8
log (2.4) = 8 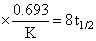
[A] = [A]0e-kt
Hence concentration of [NO0] decreases exponentially.Also, t1/2
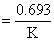 . Which is independent of concentration and t1/2 decreases with the increase of temperature.
. Which is independent of concentration and t1/2 decreases with the increase of temperature.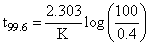
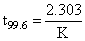 log (2.4) = 8
log (2.4) = 8 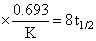
Create a free account to view solution
View Solution FreeMore Alkyl Halides and Aryl Halides Questions
What happens when CCl4 is treated with AgNO3 -...Ethanol can be prepared more easily by which reaction ?(i) CH3CH2Br + H2O → CH3CH2OH(ii) CH3CH2Br + Ag2O (in boili...What are A & B in the following reaction?...Which one of the following compounds will be most reactive for SN1 reactions:...In the given reaction : [X]...