AIPMT | 2015Basic Maths and Units and DimensionsHard
Question
Figure below shows two paths that may be taken by a gas to go from a state A to a state C.
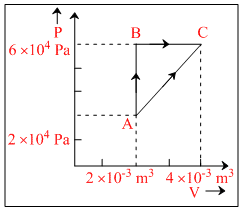
In process AB, 400 J of heat is added to the system and in process BC, 100 J of heat is added to the system. The heat absorbed by the system in the process AC will be :

In process AB, 400 J of heat is added to the system and in process BC, 100 J of heat is added to the system. The heat absorbed by the system in the process AC will be :
Options
A.500 J
B.460 J
C.300 J
D.380 J
Solution
In cyclic process ABCA,
ᐃUcyclic = 0
Qcyclic = Wcyclic
QAB + QBC + QCA = closed loop area.
400 + 100 + QCA =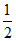 × (2 × 10-3) × 4 × 104
× (2 × 10-3) × 4 × 104
400 + 100 − QAC = 40
QAC = 460 J
Hence option (2)
ᐃUcyclic = 0
Qcyclic = Wcyclic
QAB + QBC + QCA = closed loop area.
400 + 100 + QCA =
400 + 100 − QAC = 40
QAC = 460 J
Hence option (2)
Create a free account to view solution
View Solution FreeTopic: Basic Maths and Units and Dimensions·Practice all Basic Maths and Units and Dimensions questions
More Basic Maths and Units and Dimensions Questions
The refracting angle of a prism is A, and refractive index of the material of the prism is cot(A/2). The angle of minimu...A projectional can have the same range for two angles of projection. If h1 and h2 are maximum heigh when the range in th...A body of mass m slides down an incline and reaches the bottom with a velocity V. If the same mass were in form of ring ...A man of mass 60 kg is standing on a platform of mass 40 kg as shown in figure then what force man should apply on rope ...On a smith plane surface (figure)two block A and B are accelerated up by applying aforce 15 N on A. If mass of B twice t...