AIPMT | 2015s-Block ElementsHard
Question
Solubility of the alkaline earth′s metal sulphates in water decreases in the sequence :-
Options
A.Ca > Sr > Ba > Mg
B.Sr > Ca > Mg > Ba
C.Ba > Mg > Sr > Ca
D.Mg > Ca > Sr > Ba
Solution
Due to very small size of Mg+2, Mg+2 shows maximum hydration energy.
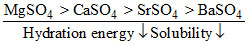
Create a free account to view solution
View Solution FreeMore s-Block Elements Questions
Which does not exist in solid state....The set representing the correct order of first ionisation potential is :...Which one of the alkaline earth metals shows some anomalous behavior and has sameelectronegative as aluminum?...Which is correct statement...The first ionisation energies of alkaline earth metal are higher than those of the alkali metals. This is because:...