PolymerHard
Question
The freezing point (in oC) of a solution containing 0.1 g of K3[ Fe(CN)6 ] (Mol. Wt. 329) in 100 g of water (Kf = 1.86 K kg mol-1) is
Options
A.- 2.3 × 10-2
B.- 5.7 × 10-2
C.- 5.7 × 10-3
D.- 1.2 × 10-2
Solution
K3[Fe(CN)6] → 3K+ + [Fe(CN)6]3-
i = 4
ᐃTf = Kf × i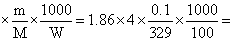 2.3 × 10-2
2.3 × 10-2
T′f = - 2.3 × 10-2
i = 4
ᐃTf = Kf × i
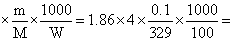 2.3 × 10-2
2.3 × 10-2 T′f = - 2.3 × 10-2
Create a free account to view solution
View Solution FreeMore Polymer Questions
In organic compounds P is estimated as-...Boiling point of a liquid can be increased by-...When methyl D-glucopyranoside is treated with HIO4 its number of mole consumed per mole of the sugare is -...Cyanocobalamin is the chemical name of:...Characters are transferred from parents to offspring through-...