Alcohol, Phenol and EtherHard
Question
Which of the following statements is not valid for oxoacids of phosphorus ?
Options
A.Orthophosphoric acid is used in the manufacture of triple super phosphate
B.Hypophosphorus acid is a diprotic acid
C.All oxoacids contain tetrahedral four coordinated phosphorus
D.All oxoacids contain at least on P = O unit and one P - OH group
Solution
Hypophosphorus acid, H3PO2 , has the following structure.
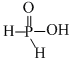
As it contains only one replaceable H-atom (that is attached with O, not with P directly) so it is a monoprotic acid.

As it contains only one replaceable H-atom (that is attached with O, not with P directly) so it is a monoprotic acid.
Create a free account to view solution
View Solution Free