Alcohol, Phenol and EtherHard
Question
Lucas test is used to make distinction between 1o, 2o and 3o alcohols
ROH +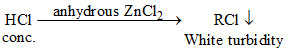 + H2O
+ H2O
This shows that
ROH +
This shows that
Options
A.ROH behaves as a base
B.greater the value of pKa (alcohol), greater the reactivity with conc. HCl and thus sooner the formation of white turbidity.
C.alcohol which reacts fastest with Na metal, will give turbidity at fastest rate
D.alcohol which gives red colour during Victor Mayor test, will give turbidity at slower rate then those giving blue or white colour during Victor Mayor test.
More Alcohol, Phenol and Ether Questions
Phenol is more acidic than cyclohexanol because -...[X] here ′X′ is:...Benzene diazonium chloride on reaction with phenol in weakly basic medium gives :...The major product obtained on interaction of phenol with sodium hydroxide and carbon dioxide is :...Match structures given in list I with names given in list II and then select the correct answer from the codes given bel...