Basic Maths and Units and DimensionsHard
Question
Standard enthalpy of vaporisation ᐃvapHo for water at 100oC is 40.66 kJ mol-1. The internal energy of vaporisation of water at 100oC (in kJ mol-1) is
(Assume water vapour to behave like an ideal gas).
(Assume water vapour to behave like an ideal gas).
Options
A.+ 37.56
B.- 43.76
C.+ 43.76
D.- 40.66
Solution
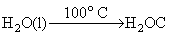
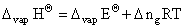
For the above reqaction,
ᐃ ng = np - nr = 1 - 0 = 1
∴ 40.66 kJ mol-1 =
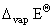 + 1 × 8.314 × 10-3 × 373
+ 1 × 8.314 × 10-3 × 373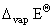 = 40.66 kJ mol-1 - 3.1 kJ mol-1
= 40.66 kJ mol-1 - 3.1 kJ mol-1= + 37.56 kJ mol-1
Create a free account to view solution
View Solution FreeTopic: Basic Maths and Units and Dimensions·Practice all Basic Maths and Units and Dimensions questions
More Basic Maths and Units and Dimensions Questions
A ray of light is incident on an equilateral glass prism (μ = √3) and moves parallel to the base of the prism...What should be the maximum acceptance angle at the air-core interface of an optical fibre if n1 and n2 are the refractiv...The co-ordinates of a moving particle at any time t are given by x = αt3 and y = βt3. The speed to the particl...A body has speed V, 2V and 3V in first 1/3 of distance S, seconds 1/3 of S and third 1/3 of S respectively. Its average ...Liquid rises to a height of 2cm in a capillary tube. The angle of contact between the solid and the liquid is zero. The ...