ThermodynamicsHard
Question
A gas undergoes an adiabatic process in which pressure becomes 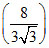 times and volume become
times and volume become 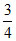 of initial volume. If initial absolute temperature was T, the final temperature is
of initial volume. If initial absolute temperature was T, the final temperature is
Options
A.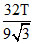
B.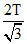
C.T3/2
D.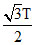
Solution
For adiabatic processP1V1γ = P2V2γ ⇒ P0V0γ = 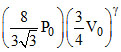 ...(i)
...(i)
Also, T1V1γ-1 = T2V2γ-1 ⇒ TV0γ-1 = T2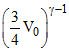 ...(ii)
...(ii)
Solving (i) & (ii), T2 =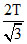
Also, T1V1γ-1 = T2V2γ-1 ⇒ TV0γ-1 = T2
Solving (i) & (ii), T2 =
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The amount of heat required to convert 1 gm of ice at 0oC into steam at 100oC, is...The wavelength of maximum energy released during an atomic explosin was 2.93 × 10-10m. The maximum temperature atta...One mole of an ideal atomic gas is heated at a constant pressure of one atmosphere from 0oC to 100oC. Then the change in...The unit of gas constant R is :-...Fraunh offer line the solar system is an example of :...