Carbonyl CompoundHard
Question
The vapour pressure of acetone at 20oC is 185 torr. When 1.2 g of non-volatile substance was dissolved in 100 g of acetone at 20oC, its vapour pressure was 183 torr. The molar mass (g mol−) of the substance is :
Options
A.128
B.488
C.32
D.64
Solution
According to Raoult′s law 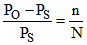
⇒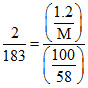
⇒ M = 64
⇒
⇒ M = 64
Create a free account to view solution
View Solution Free