d and f-Block ElementsHard
Question
The color of KMnO4 is due to :
Options
A.L → M charge transfer transition
B.σ − σ* transition
C.M → L charge transfer transition
D.d − d transition
Solution
KMnO4 = Dark Purple (Mn+7 = 3d0 configuration)
*Purple coloration is due to Ligand to metal charge transfer spectra.
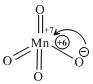
* During charge transfer oxidation state of Mn is temporary reduced to Mn+6
*Purple coloration is due to Ligand to metal charge transfer spectra.

* During charge transfer oxidation state of Mn is temporary reduced to Mn+6
Create a free account to view solution
View Solution FreeMore d and f-Block Elements Questions
An organic comp. (C7H8O) dissolves in NaOH and gives a characteristic colour with FeCl3. On treatment with bromine water...An ion 25Mnx+ has the magnetic moment of 4.9 BM. The value of x is?...Which one is paramagnetic :-...Larger number of oxidation states are exhibited by the actinoids than those by the lanthanoids, the main reason being...A first row transition metal (M) does not liberate $H_{2}$ gas from dilute HCl .1 mol of aqueous solution of ${MSO}_{4}$...