p-Block elementsHard
Question
The unbalanced chemical reactions given in List-I show missing reagent or condition (?) which are provided in List-II. Match List-I with List-II and select the correct answer using the code given below the lists :
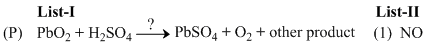

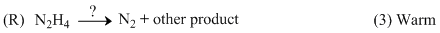

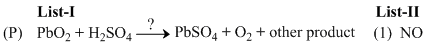

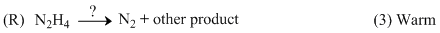

Options
A.P → 4, Q → 2, R → 3, S → 1
B.P → 3, Q → 2, R → 1, S → 4
C.P → 1, Q → 4, R → 2, S → 3
D.P → 3, Q → 4, R → 2, S → 1
Solution
(P) PbO2 + H2SO4 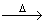 PbSO4 + O2 + 2H2O
PbSO4 + O2 + 2H2O
Other product = 2H2O
? = ᐃ
(Q) Na2S2O3 + 5H2O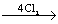 2NaHSO4 + 8 HCl
2NaHSO4 + 8 HCl
other product = 8 HCl
? = Cl2
(R) N2H4 + 2I2 → N2 + 4HI
? = I2
Other product = 4HI
(S) XeF2 + NO → Xe + NOF
Other product = NOF
? = NO
Hence answer is (D)
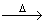 PbSO4 + O2 + 2H2O
PbSO4 + O2 + 2H2OOther product = 2H2O
? = ᐃ
(Q) Na2S2O3 + 5H2O
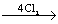 2NaHSO4 + 8 HCl
2NaHSO4 + 8 HClother product = 8 HCl
? = Cl2
(R) N2H4 + 2I2 → N2 + 4HI
? = I2
Other product = 4HI
(S) XeF2 + NO → Xe + NOF
Other product = NOF
? = NO
Hence answer is (D)
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
Which of the following possess the highest bond energy ?...Which of the following compounds does give N2 on heating?...Phosphorus trichloride, PCl3 undergoes, hydrolysis at room temperature to produce an oxoacid. It has the formula :...The correct statement(s) related to allotropes of carbon is/are...Bleaching of a fabric cloth is done using A and excess of chlorine is removed using B. A and B are :...