JEE Advanced | 2013Carbonyl CompoundHard
Question
After completion of the reactions (I and II), the organic compound(s) in the reaction mixtures is(are)
Reaction I :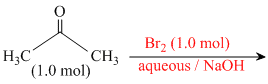
Reaction II :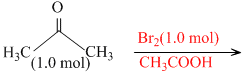
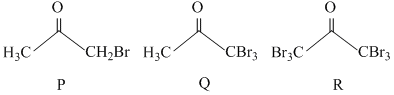
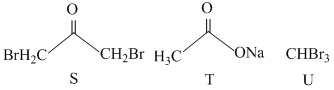
Reaction I :
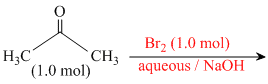
Reaction II :
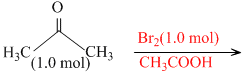

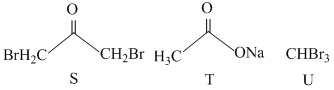
Options
A.Reaction I : P and Reaction II : P
B.Reaction I : U, acetone and Reaction II : Q acetone
C.Reaction I : T, U, acetone and Reaction II : P
D.Reaction I : R, acetone and Reaction II : S acetone
Solution
Reaction I : 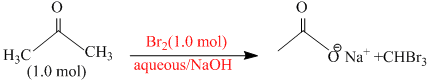
Mechanism
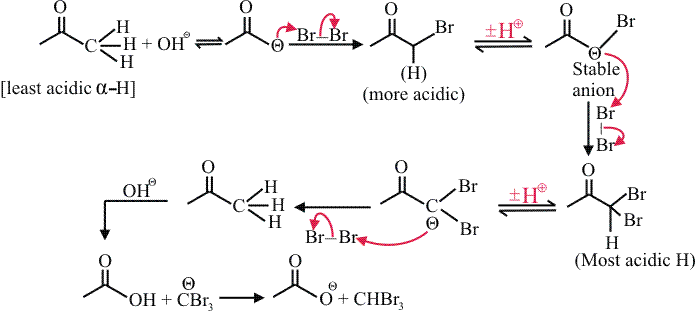
In basic medium halogenation dose not stop with replacement of just one hydrogen and poly halogenation takes place because a-haloketones are more reactive towards base and haloform reaction takes placeIn above reaction Br2 is limiting agents.
Reaction II :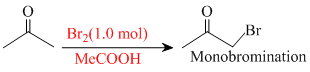

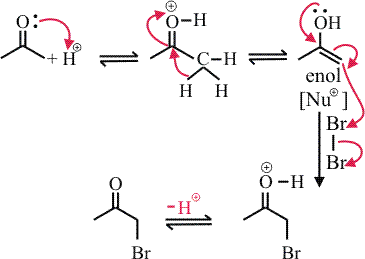
Mechanism :
Further bromination is less favourable because of less amount of Br2
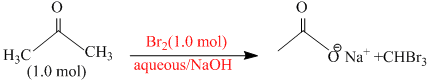
Mechanism

In basic medium halogenation dose not stop with replacement of just one hydrogen and poly halogenation takes place because a-haloketones are more reactive towards base and haloform reaction takes placeIn above reaction Br2 is limiting agents.
Reaction II :


Mechanism :

Further bromination is less favourable because of less amount of Br2
Create a free account to view solution
View Solution Free