JEE Advanced | 2013MetallurgyHard
Question
The carbon-based reduction method is NOT used for the extraction of
Options
A.tin from SnO2
B.Iron from Fe2O3
C.aluminium from Al2O3
D.magnesium from MgCO3.CaCO3
Solution
(A) 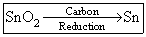
SnO2 + 2C → Sn + 2CO ↑
SnO2 + 2CO → Sn + 2CO2 ↑
(B)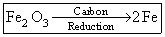
Fe2O3 + 3C → 2Fe + 3CO ↑
Fe2O3 + 3CO → 2Fe + 3CO2 ↑
(C)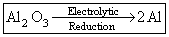
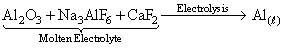
(D)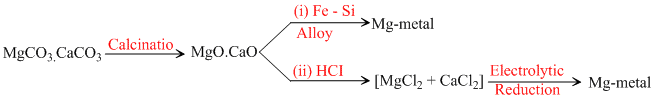
In no process carbon is used as reducing agent.

SnO2 + 2C → Sn + 2CO ↑
SnO2 + 2CO → Sn + 2CO2 ↑
(B)

Fe2O3 + 3C → 2Fe + 3CO ↑
Fe2O3 + 3CO → 2Fe + 3CO2 ↑
(C)

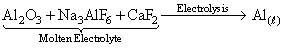
(D)

In no process carbon is used as reducing agent.
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
Which one of the following reactions occurs during smelting in the reduction zone at lower temperature (in iron metallur...Choose the correct option using the code regarding roasting process.(I) It is the process of heating the ore in air in a...Poling process is used for the refining of :...Match the reactions taking place in blast furnace with temperature - range of operations Column - I Column - II(a) C + C...Poling process is used for :...