Aromatic HydrocarbonsHard
Question
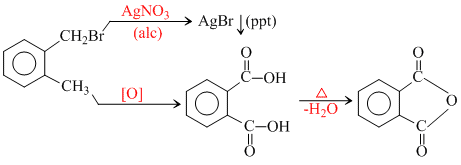
Compound (A), C8H9Br, gives a white precipitate when warmed with alcoholic AgNO3. Oxidation of (A) gives an acid (B), C8H6O4. (B) easily forms anhydride on heating. Identify the compound (A) :
Options
A.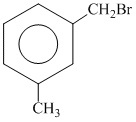

B.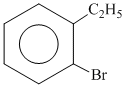

C.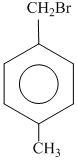

D.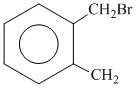

Solution
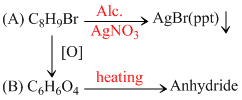
since B on heating gives anhydride so B is 1,2dicarboxylic acid. It contains two Benzylic carbon at 1,2 position so on oxidation it produces 1, 2-dicarboxylic acid
Create a free account to view solution
View Solution FreeMore Aromatic Hydrocarbons Questions
Given below are two statements:Statement I : Benzene is nitrated to give nitrobenzene, which on further treatment with${...Of the species PhSH, , and he meta-substituted product is obtained from...Polystyrene is polymer of −...The reactivity of compound Z with different halogens under appropriate conditions is given below : The observed pattern ...Heating a mixture of sodium benzoate and soda lime gives -...