p-Block elementsHard
Question
Which of the following is the wrong statement?
Options
A.ONCl and ONO- are not isoelectronic
B.O3 molecule is bent
C.Ozone is violet-black in solid state
D.Ozone is diamagnetic gas
Solution
(i) Some times, two species having samevalence shell electrons are considered asisolectronic species. Accordingly ONCl and ONO- are isoelectronic to each other.
[Otherwise all four statements are correct and no answer is available in the given options].
(ii)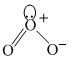 sp2, bent shaped
sp2, bent shaped
(iii)
O3(g) = Blue
O3(l) = Dark blue
O3 (s) = violet - black
(iv) All electrons are paired in structure of O3.Hence it is diamagnetic.
[Otherwise all four statements are correct and no answer is available in the given options].
(ii)
 sp2, bent shaped
sp2, bent shaped(iii)
O3(g) = Blue
O3(l) = Dark blue
O3 (s) = violet - black
(iv) All electrons are paired in structure of O3.Hence it is diamagnetic.
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
The decrease in stability of higher oxidation state in p-block with increasing atomic number is due to :...Which of the following statements is not valid for oxoacids of phosphorus?...In group 15, the melting points of the elements :...Which of the following compound can be used for oxygenating the air in submarines :...The aqueous solution of hydrogen peroxide :...