Digestion and AbsorptionHard
Question
A mixture of potassium chlorate, oxalic acid and sulphuric acid is heated. During the reaction which element undergoes maximum change in the oxidation number ?
Options
A.S
B.H
C.Cl
D.C
Solution
When a mixture of potassium chlorate, oxalic acid and sulphuric acid is heated, the following reaction occurs :
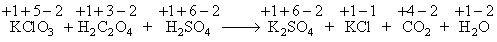
Thus, Cl is the element which undergoes maximum change in the oxidation state.
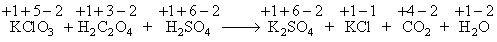
Thus, Cl is the element which undergoes maximum change in the oxidation state.
Create a free account to view solution
View Solution FreeMore Digestion and Absorption Questions
Which one of the following is the correct pairing of the site of action and the substrate of rennin...The number of salivary glands in man is -...In mammals the teeth are(i) of different type (ii) Embedded in the cup-like socket of the jaw bones (iii) Only two set, ...The vitamin-C or ascorbic acid prevents...Pernicious anaemia is caused due to the deficiency of...