Analytical ChemistryHard
Question
Bond order of 1.5 is shown by
Options
A.O2+
B.O2-
C.O22-
D.O2
Solution
(a) MO configuration of O2+ ( 8 + 8 - 1 = 15)
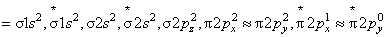
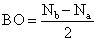
where, Nb = number of electrons in bounding molecular orbital
Na = number of electrons in anti-bounding molecular orbital
∴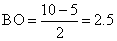
Similarly,
O2- ( 8 + 8 + 1 = 17 )
so BO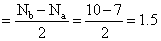
(c)O22- ( 8 + 8 + 2 = 18 )
BO
(d) O2( 8 + 8 = 16 )
BO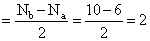
Thus,O2- shows the bond order 1.5.
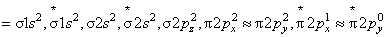
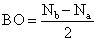
where, Nb = number of electrons in bounding molecular orbital
Na = number of electrons in anti-bounding molecular orbital
∴
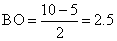
Similarly,
O2- ( 8 + 8 + 1 = 17 )
so BO
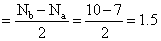
(c)O22- ( 8 + 8 + 2 = 18 )
BO

(d) O2( 8 + 8 = 16 )
BO
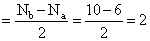
Thus,O2- shows the bond order 1.5.
Create a free account to view solution
View Solution FreeMore Analytical Chemistry Questions
A solution of a salt in concentrated sulphuric acid H2SO4 acid produced a deep blue colour with starch iodide solution. ...Which of the following statements is/are true ?...Hg2I2↓ (green) productsWhich of the following statement is correct with respect to the products ?...Which one of the following salts will produce clear and transparent original solution in 2M HCl ?...Ferric alum gives deep red colour with NH4SCN due to the formation of :...