Math miscellaneousHard
Question
In which of the folowing compounds, nitrogen exhibits highest oxidation state ?
Options
A.N2H4
B.NH3
C.N3H
D.NH2OH
Solution
Let the oxidation state of nitrogen in the given compounds be x.]
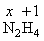
2( x ) + ( +1 )4 = 0
2x = - 4
∴ x = - 2
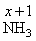
x + ( +1 )3 = 0
∴ x = - 3
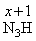
( x )3 + ( +1 ) = 0
3x = - 1
∴ x = - 1/3
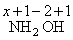
x + ( + 1 )2 + ( - 2 ) + ( + 1 ) = 0
x + 2 - 2 + 1 = 0
x + 1 = 0
∴ x = - 1
Thus, oxidation state of nitrogen is highest in N3H.
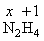
2( x ) + ( +1 )4 = 0
2x = - 4
∴ x = - 2
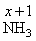
x + ( +1 )3 = 0
∴ x = - 3
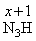
( x )3 + ( +1 ) = 0
3x = - 1
∴ x = - 1/3
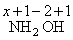
x + ( + 1 )2 + ( - 2 ) + ( + 1 ) = 0
x + 2 - 2 + 1 = 0
x + 1 = 0
∴ x = - 1
Thus, oxidation state of nitrogen is highest in N3H.
Create a free account to view solution
View Solution FreeMore Math miscellaneous Questions
Let f(x) be differentiable on the interval (0, ∞) such that f(1) = 1, and for each x > 0. Then f(x) is...How many real solutions does the equation x7 + 14x55+ 16x3 + 30x - 560 = 0 have?...Coefficient of x10 in the expansion of (1 + x)9 ln (1 + x), is (where |x|...The value of the integral, is......