Solid StateHard
Question
A metal has a fcc lattice. The edge length of the unit cell is 404 pm. The density of the metal is 2.72 gm-cm-3. The molar mass of the metal is :-
Options
A.20 gm- mol-1
B.40 gm- mol-1
C.30 gm- mol-1
D.28 gm- mol-1
Solution
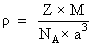 for FCC, Z = 4
for FCC, Z = 4a = 404 pm = 404 × 10-10 cm.
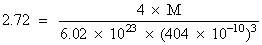
M = 27 gmol-1
Create a free account to view solution
View Solution FreeMore Solid State Questions
Copper crystallizes in fcc with a unit cell length of 361 pm. What is the radius of copper atom?...The distance between adjacent oppositely charged ions in rubidium chloride is 328.5 pm, in potassium chloride is 313.9 p...Aluminum metal has a density of 2.72 g/cm3 and crystallizes in a lattice with an edge length of 404 pm.Which of the foll...MgO exists in a rock salt type unit cell. Each Mg2+ ion will be in contact with...TiO2 is a well-known example of...