Aromatic HydrocarbonsHard
Question
Reaction by which Benzaldehyde cannot be prepared :-
Options
A.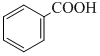 + Zn/Hg and conc. HCl
+ Zn/Hg and conc. HCl
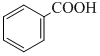 + Zn/Hg and conc. HCl
+ Zn/Hg and conc. HClB.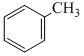 + CrO2Cl2 in CS2 followed by H3O+
+ CrO2Cl2 in CS2 followed by H3O+
 + CrO2Cl2 in CS2 followed by H3O+
+ CrO2Cl2 in CS2 followed by H3O+C.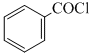 + H2 in presence of ( Pd+BaSO4)
+ H2 in presence of ( Pd+BaSO4)
 + H2 in presence of ( Pd+BaSO4)
+ H2 in presence of ( Pd+BaSO4)D.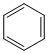 + CO + HCl in presence of anhydrous AlCl3
+ CO + HCl in presence of anhydrous AlCl3
 + CO + HCl in presence of anhydrous AlCl3
+ CO + HCl in presence of anhydrous AlCl3 Solution
In presence of Zn - Hg and conc. HCl reduction is useful specially for aldehyde and ketone but carboxylic group remains uneffected








Create a free account to view solution
View Solution FreeMore Aromatic Hydrocarbons Questions
Ethyl benzene is obtained from benzene and ethyl bromide in presence of -...Toluene, when treated with Br2 / Fe, gives p-dromotoluene as the major product because the CH2 group :...The reaction of toluene with chlorine in the presence of ferric chloride gives predominantly-...Which of the following compounds reacts slower than benzene in electrophilic bromination -...In the chlorination of benzene with Cl2 in the presence of anhydrous FeCl3, the electrophilic species that attacks the b...