Work, Power and EnergyHard
Question
A gas at pressure 6 × 105 N/m2 and volume 1m3 and its pressure falls to 4 × 105 N/m2. When its volume is 3m3. Given that the indicator diagram is a straight line, work done by the system is :-
Options
A.6 × 105 J
B.3 × 105 J
C.4 × 105 J
D.10 × 105 J
Solution
W = 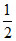 × [6 × 105 + 4 × 105] × 2
× [6 × 105 + 4 × 105] × 2
W = 10 × 105 N/m2
W = 10 × 105 N/m2
Create a free account to view solution
View Solution FreeMore Work, Power and Energy Questions
Force F on a particle moving in a straight line varies with distance d as shown in figure.The work done on the particle ...A particle is moving with a uniform speed in a circular orbit of radius R in a central force inversely proportional to t...A 10 kg satellite completes one revolution around the earth at a height of 100 km in 108 minutes. The work done by the g...In the nuclear fusion reaction, given that the repulsive potential energy between the two nuclei is −7.7 × 10-14 ...A gun of mass 10 kg fires four bullets per second. The mass of each bullet is 20 g and the velocity of the bullet when i...