p-Block elementsHard
Question
Which of these is not a monomer for a high molecular mass silicone polymer ?
Options
A.PbSiCl3
B.MeSiCl3
C.Me2SiCl2
D.Me3SiCl
Solution
Silicons are organo-silicon polymer containing 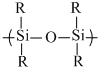 linkage. Since Me3SiCl contain only one Cl therefore it can′t form high molecular mass silicon polymer
linkage. Since Me3SiCl contain only one Cl therefore it can′t form high molecular mass silicon polymer
It act a chain terminating organo silane
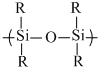 linkage. Since Me3SiCl contain only one Cl therefore it can′t form high molecular mass silicon polymer
linkage. Since Me3SiCl contain only one Cl therefore it can′t form high molecular mass silicon polymerIt act a chain terminating organo silane
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
One mole of calcium phosphide on reaction with excess water gives :...B(OH)3 + NaOH NaBO2 + Na[B(OH)4] + H2OHow can this reaction is made to proceed in forward direction?...Which of the following is the correct statement for red lead ?...Sulphuric acid acts as...Sodium nitrate decomposes above 800oC to give :...