Biology miscellaneousHard
Question
In a reaction, A + B → Product, rate is doubled when the concentration of B is doubled, and rate increases by a factor of 8 when the concentration of both the reactants (A and B) are doubled. Rate law for the reaction can be written as
Options
A.Rate = k [ A ] [ B ]2
B.Rate = k [ A ]2 [ B ]2
C.Rate = k [ A ] [ B ]
D.Rate = k [ A ]2 [ B ]
Solution
Let the order of reaction with respect to A and B is x and y respectivel. So, the rate law can be given as
R = k [ A ]X [ B ]Y ......(i)
When the concentration of only B is doubled the rate is doubled, so
R1 = k [ A ]X [ B ]X = 2R ......(ii)
If concentration of both the reactents A and B are doubled, the rate increases by a factor of 8, so
R′′ = k [ A ]X [ B ]y = 8R ......(iii)
⇒ k 2x 2y [ A ]X [ B ]y = 2R ......(iv)
From Eqs. (i) and (ii), we get
⇒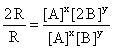
2 = 2Y
∴ y = 1
From Eqs. (i) and (ii), we get
⇒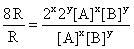
or 8 = 2x 2y
Substitution of the value of y gives,
8 = 2x 21
4 = 2x
(2)2 = 2x
∴ x = 2
Substitution of the value of x and y in Eq. (i) gives,
R = k [ A ]2 [ B ]
R = k [ A ]X [ B ]Y ......(i)
When the concentration of only B is doubled the rate is doubled, so
R1 = k [ A ]X [ B ]X = 2R ......(ii)
If concentration of both the reactents A and B are doubled, the rate increases by a factor of 8, so
R′′ = k [ A ]X [ B ]y = 8R ......(iii)
⇒ k 2x 2y [ A ]X [ B ]y = 2R ......(iv)
From Eqs. (i) and (ii), we get
⇒
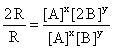
2 = 2Y
∴ y = 1
From Eqs. (i) and (ii), we get
⇒
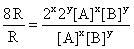
or 8 = 2x 2y
Substitution of the value of y gives,
8 = 2x 21
4 = 2x
(2)2 = 2x
∴ x = 2
Substitution of the value of x and y in Eq. (i) gives,
R = k [ A ]2 [ B ]
Create a free account to view solution
View Solution FreeMore Biology miscellaneous Questions
Synergism is shown by :-...Spindle fibres attach on to:...Which one of the following sets of items in the options 1 - 4 are correctly categorised with oneexception in it ?...How many diphyodont is present in human :-...Which of the following mRNA is inhibits by 3′-UAC GGU GAC UGG-5′ mic RNA :-...