Biology miscellaneousHard
Question
Which one of the following is an outer orbital complex and exhibits paramagnetic behaviour ?
Options
A.[ Ni(NH3)6 ]2+
B.[ Zn(NH3)6 ]2+
C.[ Cr(NH3)6 ]3+
D.[ Co(NH3)6 ]3+
Solution
Outer orbital complex utilises nd orbitals for bonding and exhibit paramagnetic behaviour, only if there present unpaired electrons.
(a) In [ Ni ( NH3)6]2+:
Ni2+ = [Ar] 3d8 4s0
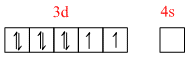
[Ni(NH3)6]2+ =
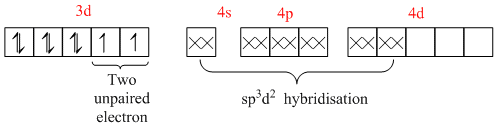
So, this is an outer orbital complex having paramagnetic character.
(b) In [ Zn ( NH3)6]2+ :
Zn2+ = [ Ar ] 3d10
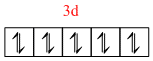
[Zn(NH3)6]2+ =
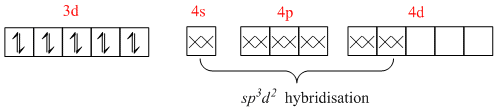
Thus, it is also an outer orbital complex but it is dimagnetic as all the electrons are paired.
(C) In [ Cr(NH3)6]3+ :
Cr3+ = [Ar]3d3
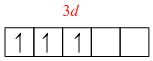
[Cr( NH3)63+ =
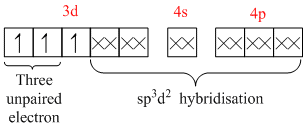
Because of the involvement of (n1)d, i.e., 3d orbital in hybridisation, it is an inner orbital complex. Its nature is paramagnetic because of the presence of three unpaired electrons.
(d) In [ Co( NH3)6]3+ :
Co3+ = [ Ar ] 3d6
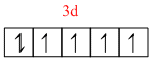
[Co(NH3)6]3+ =
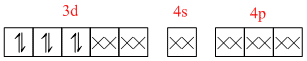
As all the electrons are paired it is dimagnetic complex.
(a) In [ Ni ( NH3)6]2+:
Ni2+ = [Ar] 3d8 4s0

[Ni(NH3)6]2+ =

So, this is an outer orbital complex having paramagnetic character.
(b) In [ Zn ( NH3)6]2+ :
Zn2+ = [ Ar ] 3d10

[Zn(NH3)6]2+ =

Thus, it is also an outer orbital complex but it is dimagnetic as all the electrons are paired.
(C) In [ Cr(NH3)6]3+ :
Cr3+ = [Ar]3d3

[Cr( NH3)63+ =

Because of the involvement of (n1)d, i.e., 3d orbital in hybridisation, it is an inner orbital complex. Its nature is paramagnetic because of the presence of three unpaired electrons.
(d) In [ Co( NH3)6]3+ :
Co3+ = [ Ar ] 3d6

[Co(NH3)6]3+ =

As all the electrons are paired it is dimagnetic complex.
Create a free account to view solution
View Solution FreeMore Biology miscellaneous Questions
Conifers are adapted to tolerate extreme environmental conditions because of...Which of the following statement about photochemical smog is wrong :-...The idea of spontaneous generation was first refused by :-...Toxic agent present in food which interfere with thyroxin synthesis lead to the developement of...In B-DNA molecule length of one complete turn is :-...