General Organic ChemistryHard
Question
The radical, 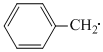 is aromatic because it has :-
is aromatic because it has :-
 is aromatic because it has :-
is aromatic because it has :-Options
A.6p-orbitals and 7 unpaired electrons
B.6p-orbitals and 6 unpaired electrons
C.7p-orbitals and 6 unpaired electrons
D.7p-orbitals and 7 unpaired electrons
Solution
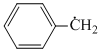
Benzyl free radical is aromatic as per Huckel′s rule it has 6π electrons present in p-orbital of carbon atoms involved in formation of benzene ring (Aromatic nature).
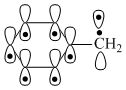 ,
,6 π electrons and 6 p orbitals
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
Arrange Decreasing order of basicityCI− RCOO− OH− RO− NH I II III IV V...The synthesis of alkyl fluoride is best accomplished by :...Methyl - α - D - glucoside and methyl - β - D - glucoside are :...Which of the following is an electrophilic reagent ?...In the reaction the electrophile involved is...