Basic Maths and Units and DimensionsHard
Question
Suppose ideal gas equation follows VP3 constant. Initial temperature and volume of the gas are T and V respectively if gas expands to 27V then its temperature will become :-
Options
A.T
B.9T
C.27 T
D.T/9
Solution
VP3 = constant = k ⇒ P = 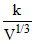
Also PV = μRT ⇒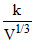 .V = μRT
.V = μRT
⇒ v2/3 =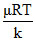 Hence
Hence 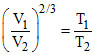
⇒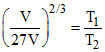 ⇒ T2 = 9 T
⇒ T2 = 9 T
Also PV = μRT ⇒
⇒ v2/3 =
⇒
Create a free account to view solution
View Solution FreeTopic: Basic Maths and Units and Dimensions·Practice all Basic Maths and Units and Dimensions questions
More Basic Maths and Units and Dimensions Questions
On a smith plane surface (figure)two block A and B are accelerated up by applying aforce 15 N on A. If mass of B twice t...Two mechanical waves, y1 = 2 sin 2π (50 t - 2x) & y2 = 4 sin 2π (ax + 100 t) propagate in a medium with same s...Consider a neutral conducting sphere. A positive point charge is placed outside the sphere. The net charge on the sphere...A wooden block is floating on the surface of water with its (1/4)th portion outside water. Its relative density and appa...The matrials sutiable for making electromagnets should have...