Alcohol, Phenol and EtherHard
Question
At 323 K, the vapour pressure in mm of Hg of a methanol-ethanol solution is represented by the equation P = 120 XA + 140, where XA is the mole-fraction of methanol. Then the value of 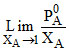 is :-
is :-
Options
A.250 mm
B.140 mm
C.260 mm
D.20 mm
More Alcohol, Phenol and Ether Questions
Among the following sets of reactants which one produces anisole?...Benzene diazonium chloride on reaction with phenol in weakly basic medium gives :...Phenol with Hinsberg′s reagent gives...The product obtained by the reaction of phenol with benzene diazonium chloride is -...Products form by following reactions are...