Benzaldehyde and Benzoic AcidHard
Question
The heat of combustion of benzoic acid at constant volume is - 321.3 kJ at 27oC. The heat of combustion at constant pressure is. (R = 8.314 JK-1 mol-1). Reaction is :
C6H5COOH(l) +15/2 O2(g) → 7CO2(g) + 3H2O(l)
C6H5COOH(l) +15/2 O2(g) → 7CO2(g) + 3H2O(l)
Options
A.-321.3 kJ
B.-322.54 kJ
C.+322.54 kJ
D.+321.3 kJ
Solution
ᐃE = qv = - 321.3 kJ/mol
ᐃH = qp = ᐃE + (ᐃng)RT
C6H5COOH(l) +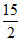 O2(g) → 7CO2 (g) + 3H2O(l)
O2(g) → 7CO2 (g) + 3H2O(l)
ᐃng = 7 -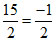
ᐃH = -321.3 +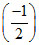 (8.314) (300) × 10-3
(8.314) (300) × 10-3
= - 322.54 kJ
ᐃH = qp = ᐃE + (ᐃng)RT
C6H5COOH(l) +
ᐃng = 7 -
ᐃH = -321.3 +
= - 322.54 kJ
Create a free account to view solution
View Solution FreeMore Benzaldehyde and Benzoic Acid Questions
Benzoic acid gives benzene on being heated with X and phenol gives benzene on being heated with Y. Therefore, X and Y ar...Which of the following compounds will react with ethanolic KCN -...C6H5CHO A + HClThe product A when reacts with the following compounds the reaction is known as Schotten Baumann reaction...Which of the following statements is correct about benzoic acid ?...Benzaldehyde condenses with acetic anhydride to give cinnamic acid in presence of -...