Solid StateHard
Question
Sodium metal adopts bcc structure, the distance between nearest sodium atom 0.368 nm. The edge length of the unit cell is :-
Options
A.0.368 nm
B.0.184 nm
C.0.575 nm
D.0.425 nm
Solution
Nearest distance = 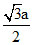
0.368 =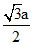
a = 0.425 nm
0.368 =
a = 0.425 nm
Create a free account to view solution
View Solution FreeMore Solid State Questions
MgO exist in a rock-salt type unit cell. Each Mg+2 ion will be in contact with...Silver crystallises in fcc lattice. If edge length of the cell is 4.07 (Ao) and density is 10.5 g cm-3. Atomic mass of t...Which of the following are the correct axial distance and axial angles for rhombohedral system?...TiO2 is a well-known example of...Spinel is an important class of oxides consisting of two types of metal ions with the oxide ions arranged in CCP pattern...