Basic Maths and Units and DimensionsHard
Question
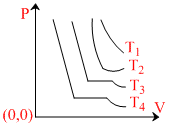
The change in volume V with respect to an increase in pressure P has been shown in the figure for a non-ideal gas at four different temperatures T1, T2, T3 and T4. The critical temperature of the gas is :-

Options
A.T1
B.T2
C.T3
D.T4
Solution
At critical temperature the horizontal portion in P-V curve almost vanishes as at temperature T2. Hence the correct answer will be(2).
Create a free account to view solution
View Solution FreeTopic: Basic Maths and Units and Dimensions·Practice all Basic Maths and Units and Dimensions questions
More Basic Maths and Units and Dimensions Questions
If the velocity of a particle is υ = At + Bt2, where A and B are constants, then the distance travelled by it betwe...Which relation is wrong...The ratio of one micron to one nanometre is...Two spherical vessel of equal volume, are connected by a narrow tube. The apparatus contains an ideal gas at one atmosph...Consider a neutral conducting sphere. A positive point charge is placed outside the sphere. The net charge on the sphere...