Work, Power and EnergyHard
Question
A vessel contains a mixture of one mole of oxygen and two moles of nitrogen at 300 K. The ratio of the average rotational kinetic energy per O2 molecule to that per N2 molecule is :-
Options
A.1 : 1
B.1 : 2
C.2 : 1
D.Depends on the moments of inertia of the two molecules
Solution
Average kinetic energy per molecule per degree of freedom = 1/2 kT. Since both the gases are diatomic and at same temperature (300K), both will have the same number of rotational degree of freedom i.e. two. Therefore, both the gases will have the same average rotational kinetic energy per molecule
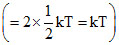 . Thus
. Thus 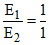
Create a free account to view solution
View Solution FreeMore Work, Power and Energy Questions
A body is moved along a straight line by a machine delivering constant power. The distance moved by the body in time t i...The kinetic energy of a body is four times its momentum. Its velocity is :-...The efficiency of an ideal heat engine working between the freezing point and boiling point of water, is :-...Radiation coming from transitions n = 2 to n = 1 of hydrogen atom falls on helium ions in n = 1 and n = 2 states. Which ...The kinetic energy needed to project a body of mass m from the earth surface (radius R) to infinity is...