Fluid MechanicsHard
Question
Assume that a drop of liquid evaporates by decrease in its surface energy, so that its temperature remains unchanged. What should be the minimum radius of the drop for this to be possible? The surface tension is T, density of liquid is ρ and L is its latent heat of vaporization.
Options
A.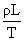
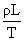
B.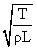
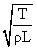
C.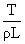
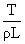
D.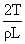
Solution
If a layer of thickness dr is evaporates then change in surface energy
= (change in surface area) T
= (d(4πr 2))T = 8πrdrT
energy required to evaporate layer of thickness
dr = (4pr2dr)ρ .L
The process of evaporation only starts only if change in surface energy is just sufficient to evaporate the water layer
⇒ (4πr2dr)Lρ = (8πrdr) T
⇒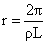
= (change in surface area) T
= (d(4πr 2))T = 8πrdrT
energy required to evaporate layer of thickness
dr = (4pr2dr)ρ .L
The process of evaporation only starts only if change in surface energy is just sufficient to evaporate the water layer
⇒ (4πr2dr)Lρ = (8πrdr) T
⇒
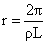
Create a free account to view solution
View Solution FreeMore Fluid Mechanics Questions
A force of 103 newton stretches the length of a hanging wire by 1 millimetre. The force required to stretch a wire of sa...A body of volume 100 c.c. is immersed completely in water contained in a jar. The weight of water and the jar before imm...For steel, the breaking stress is 6 × 106 N/m2 and the density is 8 × 103 kg/m3. The maximum length of steel w...There is no change in the volume of a wire due to change in its length on stretching. The poisson′s ratio of the m...On dipping a capillary of radius ′r′ in water, water rises upto a height H and potential energy of water is ...