ThermodynamicsHard
Question
From the following statements concerning ideal gas at any given temperature T, select the correct one (s)
Options
A.The coefficient of volume expansion at constant pressure is the same for all ideal gases
B.The verage translation kinetic energy per molecules of oxygen gas is 3 kT, k being Boltzman constant
C.The mean-free path of molecules increases with decreases in the pressure
D.In a gaseous mixture, the average translation kinetic enrgy of the molecules of each component is different
Solution
For one mole of an ideal gas pV = RT
The coefficient of volume expansion at constant pressuire is given by,
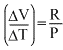 = constant [Option (a)]
= constant [Option (a)]
The average translations kinetic energy per molecule is and not 3 kT. With decrease of pressure, volume of the gas increase so its free path. [option (c)] The average translation kinetic energy of molecules is independent of their nature, so each component of the gaseous mixture will have the same value of average translational kinetic energy.
The coefficient of volume expansion at constant pressuire is given by,
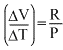 = constant [Option (a)]
= constant [Option (a)]The average translations kinetic energy per molecule is and not 3 kT. With decrease of pressure, volume of the gas increase so its free path. [option (c)] The average translation kinetic energy of molecules is independent of their nature, so each component of the gaseous mixture will have the same value of average translational kinetic energy.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The efficienty of a Carnot engine operating with reservoir tempeture of 100oC and −23oC will be :...A carnot′s engine works between temperature 772oC and 27oC, the efficienty of the engine is :...An insulated container of gas has two chambers separated by an insulating partition. One of the chambers has volume V1 a...For isometric process : -...An ideal black-body at room temperature is thrown into a furnace. It is observed that...