ThermodynamicsHard
Question
An ideal gas is taken from the state A (pressure p, volume V) to the state B (pressure p/2, volume 2V) along a straight line path in the p-V diagram. Select the correct statement from the following
Options
A.The work done by the gas in the process A to B exceeds the work that would be done by it if the system were taken from A to B along an isotherm
B.In the T-V diagram, the path AB becomes a part of a parabola
C.In the p-T diagram, the path AB becomes a part of a hyperbola
D.In going from A to B, the temperature T of the gas first increases to a maximum value and then decreases
Solution
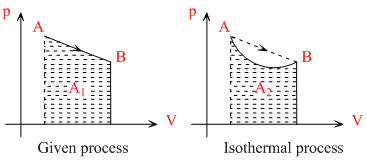
(a) Work done = Area under p-V graph
A1 > A2
∴ Wgiven process > Wisothermal process
(b) In the given process p-V equation will be of a straight line with negative slope and positive intercept i.e.,
p = - αV + β( Here α and β are positive constant)
⇒ pV = - αV2 + βV
⇒ nRT = - αV2 + βV
⇒
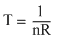 ( αV2 + βV) .....(i)
( αV2 + βV) .....(i)This is an equation of parabola in T and V.
(d)
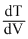 = 0 = β - 2αV
= 0 = β - 2αV⇒ V =
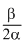
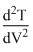 = - aαV = - ve
= - aαV = - vei.e., T has some maximum value
Now, T ∝ pV
and (pA)A = (pV)B
⇒ TA = TB
We conclude that temperatures are same at A and B and in between temperatureshas a maximum value. Therefore, in going from A to B, T will first increase to a maximum value and then decrease.
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The thermal conductivity of a rod 2.What is its thermal resistivity ?...20 litres of hydrogen gas at one atmospheric pressure is compressed at 5 atmospheric pressure, without involving any cha...A Carnot engine operating between temperatures T1 and T2 has efficiency 1. 6When T2 is lowered by 62K, its efficiency in...An amount Q of heat is added to a monoatomic ideal gas in thermodynamic process. In the process gas expands and does Q/2...Which of the following is incorrect regarding the first law of thermodynamics?...