Alcohol, Phenol and EtherHard
Question
100 g sample of methyl alcohol contains 0.002 g of water. The amount of pure methyl alcohol in terms of significant figures is reported as :-
Options
A.99.998
B.99.99 g
C.99g
D.100g
Solution
Amount of pure methyl alcohol = Amount of pure methyl alcohol = 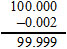
Amount should be represent in three significant figures rounding off 99.998 in three figures after rounding off amount = 100 g
Amount should be represent in three significant figures rounding off 99.998 in three figures after rounding off amount = 100 g
Create a free account to view solution
View Solution FreeMore Alcohol, Phenol and Ether Questions
The reaction 2C2H5OH (C2H5)2 O + H2Ois an example of...Which one of the statements about HOH2CCH(OH)CHO is not correct ? It...Benzyl alcohol is -...Among the following ethers, which one will produce methyl alcohol on treatment with hot concentrated HI ?...When phenol (excess) is heated with PCl5, the major product formed is -...