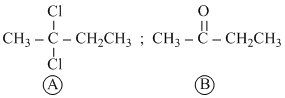Carbonyl CompoundHard
Question
Compound C4H8Cl2  on hydrolysis gives a compound C4H8O
on hydrolysis gives a compound C4H8O  which reacts with hydroxylamine and does not give any test with Tollen′s reagent. What are
which reacts with hydroxylamine and does not give any test with Tollen′s reagent. What are  and
and  respectively:-
respectively:-
 on hydrolysis gives a compound C4H8O
on hydrolysis gives a compound C4H8O  which reacts with hydroxylamine and does not give any test with Tollen′s reagent. What are
which reacts with hydroxylamine and does not give any test with Tollen′s reagent. What are  and
and  respectively:-
respectively:-Options
A.1,1-dichlorobutane and butanal
B.2,2-dichlorobutane and butanal
C.1,1-dichlorobutane and butan-2-one
D.2,2-dichlorobutane and butan-2-one
More Carbonyl Compound Questions
An organic compound reduces Tollens reagent and Fehling′s solution. It can be−...Stability of hydrates of carbonyl compounds depends on :...Fehling solution is made by mixing two separate solutions. One of which is a solution of copper sulphate only while the ...Identify ′C′ product ?...The given reactionC6H5-CHO + Br-CH2-COOC2H5 C6H5-CH=CH-COOC2H5is known as :...