p-Block elementsHard
Question
If LPG cylinder contains mixture of butane and isobutane, then the amount of oxygen that would be required for combustion of 1 kg of it will be:-
Options
A.1.8 kg
B.2.7 kg
C.4.5 kg
D.3.58 kg
Solution
C4H10 + 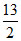 O2 → 4CO2 + 5H2O
O2 → 4CO2 + 5H2O
58 g 13 × 16g
∵ Oxygen required for combustion of 58 g butane = 13 × 16 g
∴ Oxygen required for combustion of 1 kg butane =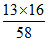 kg = 3.586 kg
kg = 3.586 kg
58 g 13 × 16g
∵ Oxygen required for combustion of 58 g butane = 13 × 16 g
∴ Oxygen required for combustion of 1 kg butane =
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
Alkaline KI is oxidised by ozone to :...NH3 can′t be obtained by :...CCl4 is inert towards hydrolysis but SiCl4 is readily hydrolysed because...The formation of O2+ [PtF6]- is the basis for the formation of xenon fluorides. This is because :...The basic strength of the hydrides of group 15 elements :...