Hydrogen and its compoundHard
Question
Which of tho following statenment (s) is (are) correct, when a mixture of NaCl and K2Cr2O7 is gently warmed with conc H2SO4 ?
Options
A.A deep red vapours is evolved
B.The vapours when passed into NaOH solution gives a yellow solution of Na2CrO4
C.Chlorine gas is evolved
D.Chromyl chloride is formed
Solution
K2Cr2O7 + 2H2SO4 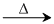 2KHSO4 + 2CrO3 + H2O
2KHSO4 + 2CrO3 + H2O
conc.
CrO3 + 2NaCl → CrO2Cl2 ↑ + H2O
Red vapours (Chromyl chloride)
CrO2Cl2 + 4NaOH → Na2CrO4 + 2NaCl + 2H2O
yellow
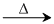 2KHSO4 + 2CrO3 + H2O
2KHSO4 + 2CrO3 + H2Oconc.
CrO3 + 2NaCl → CrO2Cl2 ↑ + H2O
Red vapours (Chromyl chloride)
CrO2Cl2 + 4NaOH → Na2CrO4 + 2NaCl + 2H2O
yellow
Create a free account to view solution
View Solution Free