Hydrogen and its compoundHard
Question
The solubility product of SrF2 in water is 8 × 10-10 calculate its solubility in 0.1M NaF aqueous solutions :-
Options
A.8 × 10-9 M
B.0.8 × 10-9 M
C.8 × 10-8 M
D.8 × 10-7 M
Solution
NaF → Na+ + F-
0.1 M 0.1 M
SrF2 Sr+2 + 2F-
1-S′ S′ 2S′
Ksp = [Sr+2] [F-]2 = (S′) (2S′ + 0.1)2
= S′(2S′2 + 0.01 + 0.4S′)
⇒ Ksp ≈ 0.01S′ ⇒ S′ =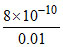 = 8 × 10-8 M
= 8 × 10-8 M
0.1 M 0.1 M
SrF2 Sr+2 + 2F-
1-S′ S′ 2S′
Ksp = [Sr+2] [F-]2 = (S′) (2S′ + 0.1)2
= S′(2S′2 + 0.01 + 0.4S′)
⇒ Ksp ≈ 0.01S′ ⇒ S′ =
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
The number of σ and π bonds present in ethane is :...Hydrogen peroxide in its reaction with KIO4 and NH2OH respectively, is acting as a...Compound having only primary hydrogen atoms is :...Iron is rendered passive by treatment with concentrated :...The solubility in water of a sparingly soluble salt AB2 is 1.0 × 10-5 mol L-1. Its solubility product number will b...