Hydrogen and its compoundHard
Question
One litre hard water contains 12.00 mg Mg2+, milliequivalents of washing soda required to remove its hardness is :- (At. wt. of Mg = 12)
Options
A.2
B.12.16
C.1 × 10-3
D.12.16 × 10-3
Solution
Milliequivalent of washing soda = meq. of Mg2+
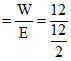 = 2 meq.
= 2 meq.
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
Among the following insecticide is :...The complex ion which has no ′d′ - electrons in the central metal atom is :...One of the constituent of German silver is :...HgCl2 and I2 both when dissolved in water containing I- ions the pair of species formed is :...Ammonium dichromate is some fireworks. The green coloured powder blown in the air is :...