Ionic EquilibriumHard
Question
Which can give Mulliken & Barkar test :-
Options
A.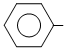 N = O
N = O
 N = O
N = OB. NO2
NO2
 NO2
NO2C. NH2
NH2
 NH2
NH2D. N
N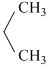
 N
N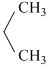
More Ionic Equilibrium Questions
The solubility product of PbI2 is 7.2 × 10−9. The maximum mass of NaI which may be added in 500 ml of 0.005 M-Pb(NO3)2 s...Which of the following buffers have a pH greater than 7 ?(a) AgOH + AgBr(b) CH3COOH + CH3COONa(c) Mg(OH)2 + Mg(OH)Cl(d) ...The pH of an aqueous solution of sodium chloride at 60o C is...A solution having hydrogen ion concentration is 0.0005 g eqvt./litre, its pOH is :...A pair of salts are given in a solution each in 0.1M concentration. Which solution has a higher pH ?...