p-Block elementsHard
Question
The correct order of acidic stregth is :
Options
A.Cl2O7 > SO2 > P4O10
B.CO2 > N2O5 > SO2
C.Na2O > MgO > Al2O3
D.K2O > CaO > MgO
Solution
On moving L → R across a pariod, the metallic character decreases while non-metallic character inceases. The nature of oxides changes from basic to acidic i.e.,why
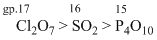
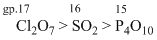
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
KF combines with HF to form KHF2. The compound contains the species :...The most abundant metal in the earth crust is :...A sulphate of a metal (A) on heating evolves two gases (B) and (C) and an oxide (D). Gas (B) turns K2Cr2O7 paper green w...Thermodynamically, the most stable form of carbon is...Ozone layer is being depleted. This is due...