p-Block elementsHard
Question
In compounds of type ECl3, where E = B, P As or Bi, the Cl - E - Cl bond angle for different E are in the order :
Options
A.B > P = As = Bi
B.B > P > As > Bi
C.B < P = As = Bi
D.B < P < As < Bi
Solution
Using different atoms assingned for E, the examples of halides are BCl3, PCl3, AsCl3 and BiCl3. In BCl3 the central B atom is sp2 hybridised and due to planar triangular geometry Cl - B - Cl angle is 120o (maximum).
In PCl3, AsCl3 and BiCl3, the central atpm is sp3 hybridised but the slight difference in angles (with slight deviation from normal tetrmal tetrahedral angle 109o28′) is due to difference in electronegativites of central atoms.
When electronegativity of central atom will be maximum, the bond pairs of E - Cl bonds will be shifted torward central atom and they will be situated closer to each other. It will lead to more bond pair-bond pair repulsion resulting in larger bond angle and vice versa.
Electromegativity of P, As and Bi follow the order :
P > As > Bi
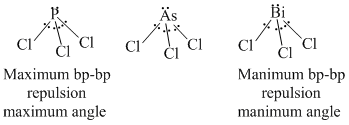
thus correct order is BCl3 > PCl3 > AsCl3 > Bi
Here it is important to note that all the three halides of P, As and Bi, the bond angle is less than 109o28′ (normal terahedral angle). It is due to lp - bp repulsion. bp-bp repulsions are not so string to exceed the Cl - E - Cl bond angle larger than 109o28′
In PCl3, AsCl3 and BiCl3, the central atpm is sp3 hybridised but the slight difference in angles (with slight deviation from normal tetrmal tetrahedral angle 109o28′) is due to difference in electronegativites of central atoms.
When electronegativity of central atom will be maximum, the bond pairs of E - Cl bonds will be shifted torward central atom and they will be situated closer to each other. It will lead to more bond pair-bond pair repulsion resulting in larger bond angle and vice versa.
Electromegativity of P, As and Bi follow the order :
P > As > Bi

thus correct order is BCl3 > PCl3 > AsCl3 > Bi
Here it is important to note that all the three halides of P, As and Bi, the bond angle is less than 109o28′ (normal terahedral angle). It is due to lp - bp repulsion. bp-bp repulsions are not so string to exceed the Cl - E - Cl bond angle larger than 109o28′
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
HBr and HI can reduce H2SO4, HCl can reduce KMnO4 and HF can reduce :...1 mol each of H3PO2, H3PO3 and H3PO4 will neutralise x mole of NaOH, y mol of Ca(OH)2 and z mol of Al(OH)3 (assuming all...27 gm of Al will react completely with how many grams of oxygen ?...The material in solar cells contains :...A gaseous substance dissolve in water giving a pale blue solution which decolourises KMnO4 and oxidises KI to I2 in acid...