p-Block elementsHard
Question
Hydrolysis of one mole of preoxodisulphuric acid produces :
Options
A.two moles of sulphuric acid
B.two moles of peroxomono sulphuric acid
C.one mole of sulphuric acid and one mole of peroxomono sulphuric acid
D.one mole of sulphuric acid, one mole of peroxomono sulphuric acid and mole of hydrogen peroxide
Solution
On hydrolysis preoxodisulphuric acid gives one mole of peroxo monosulphuric acid and one mole of sulphuric acid.
H2S2O8 + H2O → H2SO5 + H2SO4
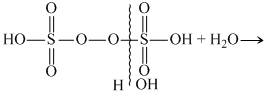
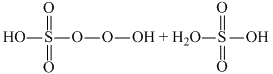
If hydrolysed completely with excess with excess of water , it gives two moles of H2SO4 and one moles of H2O2
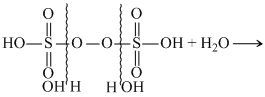 2H2SO4 + H2O2
2H2SO4 + H2O2
H2S2O8 + H2O → H2SO5 + H2SO4

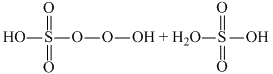
If hydrolysed completely with excess with excess of water , it gives two moles of H2SO4 and one moles of H2O2
 2H2SO4 + H2O2
2H2SO4 + H2O2Create a free account to view solution
View Solution FreeMore p-Block elements Questions
Select CORRECT statement about ozone -...For electron affinity of halogens which of the following is correct ?...When thiosulphate ion is oxidised by iodine, the new product formed is :...Which of the following product(s) is/are obtained when a mixture of NaNH2 and N2O is heated at temperature 175ºC ?...Which of the following is / are correct for group 14 elements?...