SolutionHard
Question
1.8 gm of fructose is added to 2kg of water. The freezing point of the solution is :-
(Kf = 1.86 K molality-1)
(Kf = 1.86 K molality-1)
Options
A.-1.86oC
B.0.0093oC
C.-0.0186oC
D.-0.0093oC
Solution
ᐃTf = Kf m
= 1.86 ×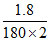 = 0.0093
= 0.0093
F.P. = 0 - 0.0093 = - 0.0093oC
= 1.86 ×
F.P. = 0 - 0.0093 = - 0.0093oC
Create a free account to view solution
View Solution FreeMore Solution Questions
The depression of freezing points of 0.05 molal aqueous solution of the following compounds are measured.1. NaCl 2. K2SO...In the depression of freezing point experiment, it is found that the :...The freezing point of equimolar aqueous solution will be highest for...Which combination of (I) vapour pressure, (II) intermolecular forces and (III) ᐃHvap (latent heat of vaporisation)...The vapour pressure of pure benzene, C6H6 at 50oC is 268 Torr. How many moles of non-volatile solute per mol of benzene ...