p-Block elementsHard
Question
Ammonia gas can be dried by :
Options
A.conc. H2SO4
B.P2O5
C.CaCl2
D.quick lime
Solution
Except CaO, NH3 reacts with all other dehydrating agents.
2NH3 + H2SO4 → (NH4)2SO4
CaCl2 + 4NH3 → CaCl4 . 4NH3
P4H10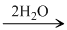 4HPO3
4HPO3 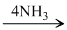 4NH4PO3
4NH4PO3
Meta phosphoric acid
2NH3 + H2SO4 → (NH4)2SO4
CaCl2 + 4NH3 → CaCl4 . 4NH3
P4H10
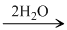 4HPO3
4HPO3 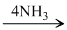 4NH4PO3
4NH4PO3 Meta phosphoric acid
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
A gas that can not be collected over water is :...The oxide which is not a reducing agent is :...Red phosphorus can be prepared from white phosphorus by :...One mole of calcium phosphide on reaction with excess water gives :...A colourless gas which burns with blue flame and reduces CuO to Cu is :...